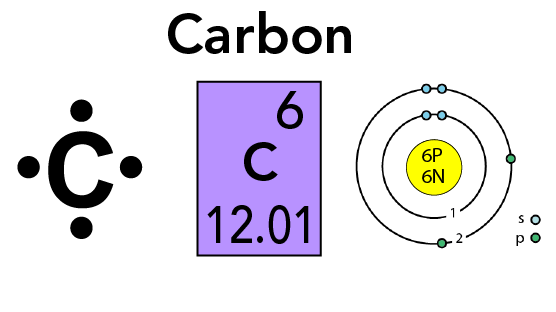
The Newest Elements on the Periodic Table

The symbols and atomic numbers for the chemical elements Nihonium, Moscovium, Ognassen and Tennessine (Dr_Micbrobe, iStockphoto)

The symbols and atomic numbers for the chemical elements Nihonium, Moscovium, Ognassen and Tennessine (Dr_Micbrobe, iStockphoto)
8.17
How does this align with my curriculum?
Curriculum Alignment
AB
11
Knowledge and Employability Science 20-4 (2006)
Unit A: Applications of Matter and Chemical Change
BC
7
Science Grade 7 (June 2016)
Big Idea: Elements consist of one type of atom, and compounds consist of atoms of different elements chemically combined.
BC
9
Science Grade 9 (June 2016)
Big Idea: The electron arrangement of atoms impacts their chemical nature.
NU
9
Knowledge and Employability Science 9 (Alberta, Revised 2009)
Unit B: Matter and Chemical Change
NU
11
Knowledge and Employability Science 20-4 (Alberta, 2006)
Unit A: Applications of Matter and Chemical Change
YT
7
Science Grade 7 (British Columbia, June 2016)
Big Idea: The electromagnetic force produces both electricity and magnetism.
YT
9
Science Grade 9 (British Columbia, June 2016)
Big Idea: The electron arrangement of atoms impacts their chemical nature.
NT
9
Knowledge and Employability Science 9 (Alberta, Revised 2009)
Unit B: Matter and Chemical Change
NT
11
Knowledge and Employability Science 20-4 (Alberta, 2006)
Unit A: Applications of Matter and Chemical Change
NT
10
Knowledge and Employability Science 10-4 (Alberta, 2006)
Unit A: Investigating Properties of Matter
BC
8
Science Grade 8 (June 2016)
Big Idea: The behaviour of matter can be explained by the kinetic molecular theory and atomic theory.