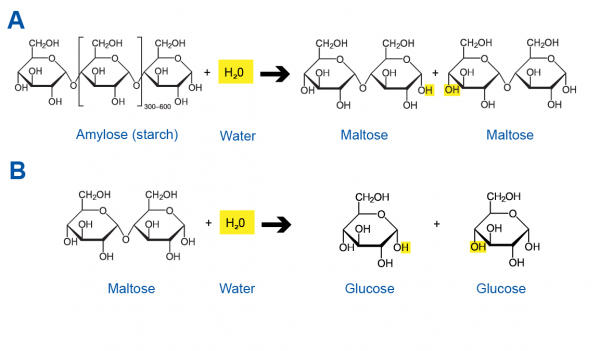
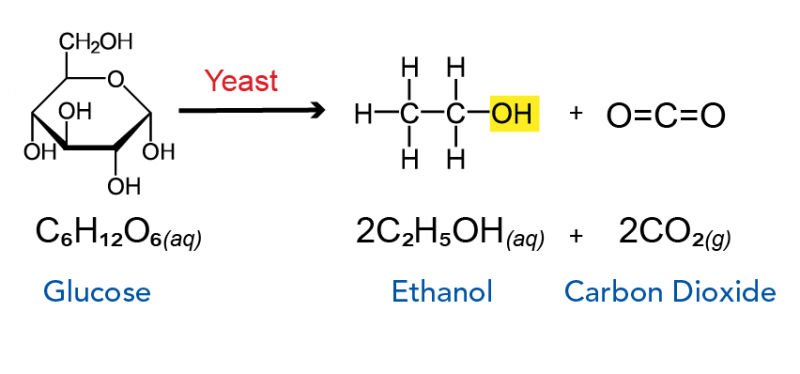
Hop To It! The Science Behind Beer

<Weighing the hops (Markus Raupach [CC BY-SA 3.0], Wikimedia Commons)

<Weighing the hops (Markus Raupach [CC BY-SA 3.0], Wikimedia Commons)
5.73
How does this align with my curriculum?
Curriculum Alignment