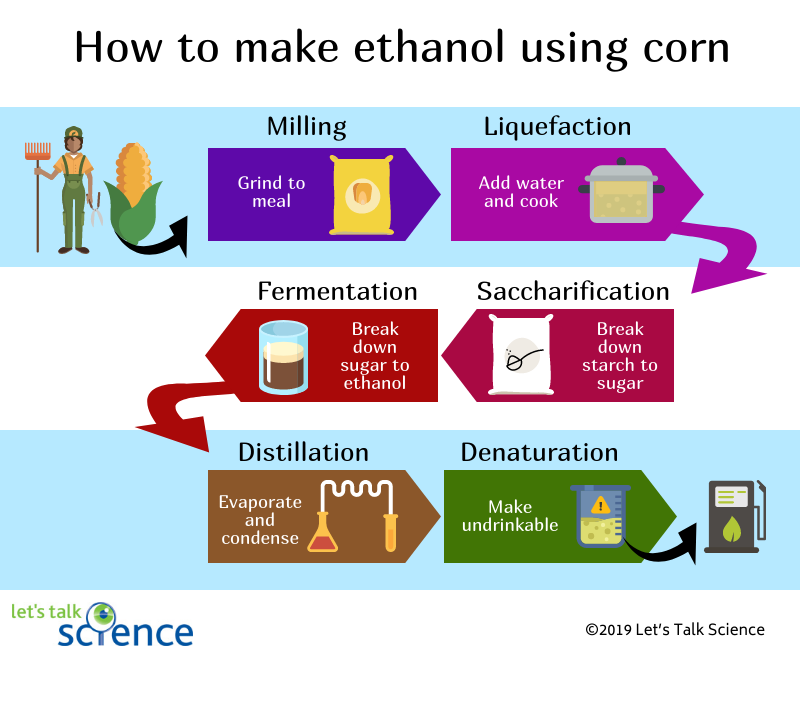
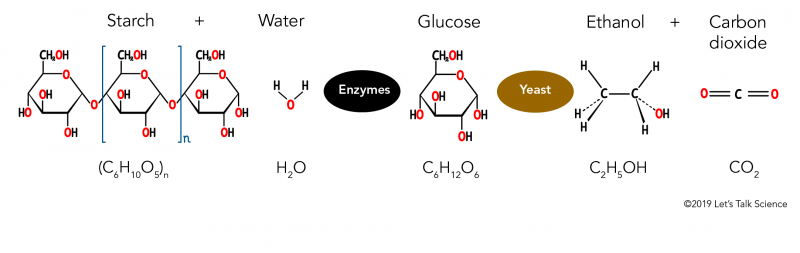
How is Ethanol Made?

Corn ethanol plant next to corn field (photosbyjim, iStockphoto)

Corn ethanol plant next to corn field (photosbyjim, iStockphoto)
8.48
How does this align with my curriculum?
BC
11
Chemistry 11 (June 2018)
Big Idea: Organic chemistry and its applications have significant implications for human health, society, and the environment.
YT
11
Chemistry 11 (British Columbia, June 2018)
Big Idea: Organic chemistry and its applications have significant implications for human health, society, and the environment.