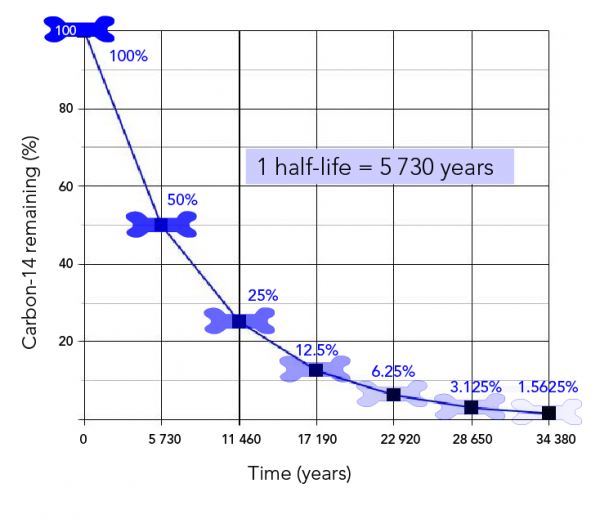
The Dating Habits of Archaeologists

Several longhouse and other stone structures on Pamiok Island, in Ungava Bay, Quebec (Lkovac, Wikimedia Commons).

Several longhouse and other stone structures on Pamiok Island, in Ungava Bay, Quebec (Lkovac, Wikimedia Commons).
8.05
How does this align with my curriculum?
Curriculum Alignment
ON
11
Chemistry, Grade 11, University (SCH3U)
Strand B: Matter, Chemical trends, and Chemical Bonding
ON
12
Earth and Space Science, Grade 12, University (SES4U)
Strand D: Recording earth’s Geological History
BC
12
Geology 12 (June 2018)
Big Idea: Earth’s geological and biological history is interpreted and inferred from information stored in rock strata and fossil evidence.