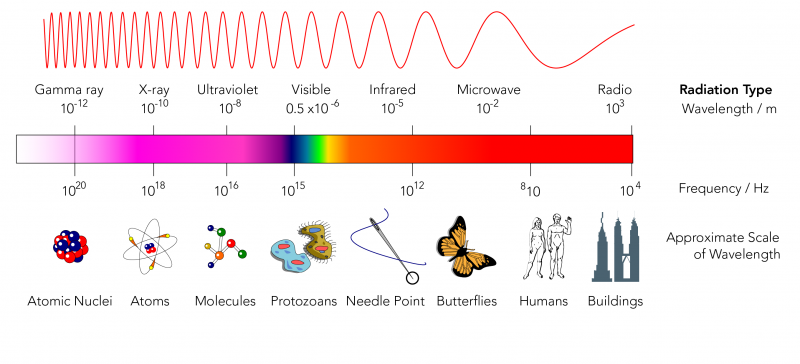
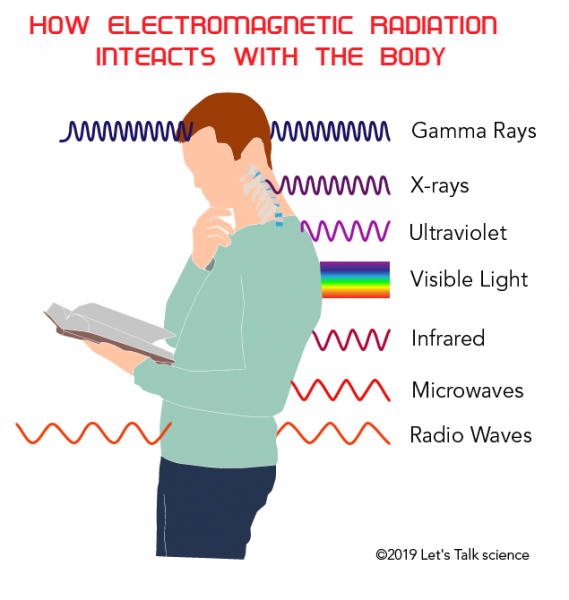
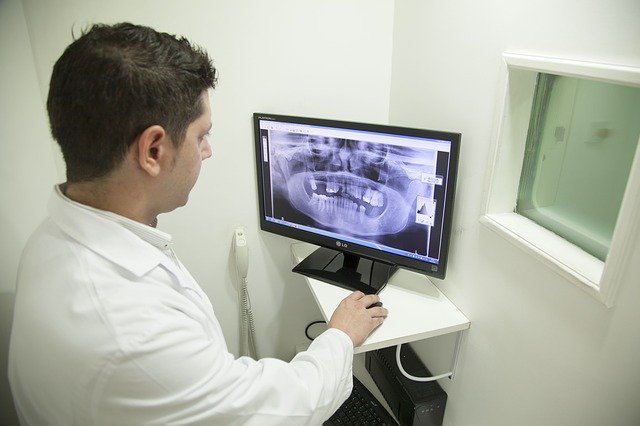
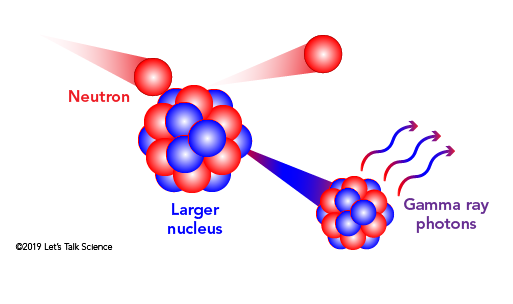
What are the Different Types of Radiation?

Artist’s interpretation of astronaut in space (isitsharp, iStockphoto)

Artist’s interpretation of astronaut in space (isitsharp, iStockphoto)
8.5
How does this align with my curriculum?
NU
8
Knowledge and Employability Science 8 (Alberta, Revised 2009)
Unit C: Light and Optical Systems
YT
8
Science Grade 8 (British Columbia, June 2016)
Big Idea: Energy can be transferred as both a particle and a wave.
NT
8
Knowledge and Employability Science 8 (Alberta, Revised 2009)
Unit C: Light and Optical Systems